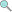3.0 credits
22.5 h + 7.5 h
1q
Teacher(s)
Robiette Raphaël ;
Riant Olivier ;
Language
Français
Main themes
This course is aimed to a synthesis of various notions related to physical organic chemistry and already introduced in the various courses from the preceding years. It also gives an introduction to some selected physico-chemical tools used in the elucidation of reaction mechanisms in organic chemistry.
The main themes are:
-Structure -activity relationships in organic chemistry
-Electronic and sterics effects
-Influence of the reaction media in organic chemistry
-Stereoelectronic effects in organic chemistry
Aims
The aim of this course is to introduce important notions and concepts selected in the field of physical organic chemistry. One of the goals of this course is to use those notions for a better understanding of reaction mechanisms in organic chemistry, the structure of reaction intermediates and transition states, and a deeper understanding of the molecular interactions which can influence chemical reactivity.
The contribution of this Teaching Unit to the development and command of the skills and learning outcomes of the programme(s) can be accessed at the end of this sheet, in the section entitled “Programmes/courses offering this Teaching Unit”.
Content
1-Structure-activity relationships: Free energy relationships. Quantitative estimation of electronic effects (Hammett and Yukawa-Tsuno equations). Application to organic reaction mechanisms. Sigma scales. Deviations to the Hammett equation. Physico-chemical methods for the measurements of the sigma parameters. Steric effects (Taft equation). Applications on literature examples.
2-Influence of the reaction media in organic chemistry: Influence of the solvent in organic chemistry. Classification of the solvents and solvents scales. Ion effects (anionic activation and electrophilic assistance). Medium effect on the reaction kinetics. Hughes-Ingold rule. Solvatation effects on reactivity.
3-Stereoelectronic effects in organic chemistry: Orbital interaction rules. Geometric restriction of the orbital overlap. Stereolectronic effects on the conformations. Anomeric effect. Stereoelectronic effects on reactivity. Effects through chemical bonds and through space. Substitution of a saturated carbon atom. Baldwin rules. SN2 reaction. Stabilisation effect on a SN2 transition state by a neighbouring group. Anchimeric assistance. Cabocations and SN1 mechanism. Reaction on a Sp2 carbon. Nucleophilic addition on a carbonyl group. Cram, Felkin-Ahn chelated Cram rules. Iminium cations and electrophilic alkenes. Intramolecular nucleophilic additions.
Other information
Background: knowledge of organic chemistry from the previous years (Bachelor of Chemistry) and CHM2140
Evaluation: written exam
Support: Books from the CHOM library, publications and databases.
The course could be partly or totally delivered by an invited lecturer.
Faculty or entity<